Alzheimer's Matters Blog
Developing Drugs
December 2, 2022
CTAD Showcases ADDF Leadership and Historic Progress in Alzheimer’s Research
The 2022 Clinical Trials on Alzheimer’s Disease (CTAD) conference has been a truly historic meeting, and the Alzheimer’s Drug Discovery Foundation (ADDF)’s role in bringing about a new era of research was evident throughout.


ADDF Impact
October 17, 2022
Celebrating Alzheimer’s Science and Scientists at the 2022 ADDF Goodes Prize Luncheon
Five previous winners of the prestigious Melvin R. Goodes Prize came together in New York City recently to honor this year’s winner, Miranda E. Orr, PhD, Assistant Professor of Gerontology and Geriatric Medicine at the Wake Forest University School of Medicine, and to discuss how their research is working toward the same goal – to conquer Alzheimer’s.


Protecting Brain Health
May 31, 2022
Can Lifestyle Interventions Combined with a Diabetes Drug Prevent Cognitive Decline?
Alzheimer’s Drug Discovery Foundation Funds Second Phase of Global Alzheimer’s Prevention FINGER TRIAL


January 7, 2022
2021 and Beyond: Research Forges Beyond Aduhelm with More Drug Approvals Expected from Diverse and Promising Pipeline
First new Alzheimer’s treatment approved in 17 years, two more get breakthrough designation for FDA review
The FDA approved aducanumab (Aduhelm®), an antibody that clears amyloid plaques from the brain, in June.


Developing Drugs
November 23, 2021
A Conversation with Our ADDF-Harrington Scholars: Studying Drug Candidates to Restore Brain Function in Alzheimer’s Patients
Earlier this year, two outstanding researchers, Paul Fish, Ph.D., and Paul Worley, M.D., were selected by the ADDF and Harrington Discovery Institute to receive ADDF-Harrington Scholar Awards.
November 19, 2021
CTAD 2021 Reflects Promising Movement in Alzheimer’s Research
The theme of the 14th Clinical Trials on Alzheimer’s Disease (CTAD) conference asked a question: Therapeutic Trials in AD: A New Hope for 2022?
Understanding Dementia
October 22, 2021
ADDF’s 22nd International Conference on Alzheimer’s Drug Discovery Pushes for Better Clinical Trials and Highlights the Importance of Biomarkers
The Alzheimer’s Drug Discovery Foundation (ADDF) hosted its 22nd International Conference on Alzheimer’s Drug Discovery on October 4-5, 2021.


Understanding Dementia
August 9, 2021
AAIC 2021 Features Several ADDF-Funded Researchers and Heavy Focus on Biomarkers
The ADDF was well represented at this year’s Alzheimer’s Association International Conference (AAIC) by our own scientific staff and our funded researchers.
Developing Drugs
July 1, 2021
The Vital Role of Patients in Alzheimer’s Research
Robust Alzheimer’s Clinical Trial Portfolio Provides More Opportunities for Patient Participation
Developing Drugs
June 11, 2021
ADDF Puts Approval of Newly FDA-Approved Alzheimer’s Drug in Context
The FDA made a landmark decision on June 7, approving aducanumab (Aduhelm™, Biogen) for the treatment of Alzheimer’s disease. Aducanumab is the first new Alzheimer’s treatment in 17 years and the first ever shown to modify the course of the disease.
Developing Drugs
June 10, 2021
A Conversation with the Scientists: Two Phase 2 Trials Exploring Novel Targets
I sat down with two outstanding ADDF-funded researchers recently to talk about the latest news on their cutting-edge work, the inspiration for their ideas, and when we can expect results from their latest trials.


ADDF Impact
January 12, 2021
2020 Year in Review: A Defining Time
This year tested us like no other, but from great challenges came innovation and creativity as we drove forward our mission to identify new therapies to prevent and treat Alzheimer’s disease.
Developing Drugs
December 1, 2020
ADDF Fosters Collaboration, Shares Expertise at CTAD 2020
The ADDF scientific affairs team and I recently participated in the 13th Clinical Trials on Alzheimer’s Disease (CTAD) conference.

ADDF Impact
August 25, 2020
ADDF Young Professionals Committee: The New Generation of Alzheimer’s Advocates
The ADDF’s Young Professionals Committee (YPC) is a group of individuals dedicated to making an impact in the fight against Alzheimer’s.


Understanding Dementia
August 19, 2020
Biomarker Blood Test on the Horizon, ADDF Biology of Aging Presentation among AAIC 2020 Highlights
The team of ADDF scientists who attended last week’s virtual Alzheimer’s Association International Conference identify their top 5 takeaways.


ADDF Impact
August 10, 2020
ADDF Venture Philanthropy Model: Taking Risks and Harnessing Rewards to Accelerate Alzheimer’s Research
The Alzheimer’s Drug Discovery Foundation’s venture philanthropy model has a singular mission: to invest money and resources in bold ideas that can speed development of drugs for Alzheimer’s.


Protecting Brain Health
August 4, 2020
Why Women Might Be at a Higher Risk for Alzheimer’s
A study published last week in Neurology, “Sex-Driven Modifiers of Alzheimer Risk,” provides new insights into why women are more than twice as likely as men to develop Alzheimer’s disease.

Developing Drugs
May 12, 2020
ADDF-Harrington Scholar Program: Bridging the Gap from Early Drug Discovery to Clinical Development
As the 2019 recipient of the ADDF-Harrington Scholar Award, Dr. Eugenia Trushina of Mayo Clinic Rochester not only receives funding, but also in-depth advisory support from pharmaceutical industry experts on the Harrington team.


Protecting Brain Health
April 3, 2020
Taking Care of Yourself and Your Loved One During COVID-19
The health of the caregiver and patient are intertwined now more than ever. Here is some helpful advice.


ADDF Impact
January 13, 2020
The Year in Review: ADDF’s Top 5 Science & Research 2019 Highlights
Over the past year, we have had many reasons to be proud of the promising work supported by the ADDF. The following is a summary of what I see as the ADDF’s most important science and research involvements of 2019. As we close out the decade, I am encouraged that we are moving closer to viable therapies and prevention strategies for Alzheimer’s disease.


Supporter Stories
January 2, 2020
Pamela Newman: An Alzheimer’s Journey, From Frustration to Revelation to Hope
By any definition, Pamela J. Newman, Ph.D. and her late husband Henry (Hank) Kates were a power couple. But when Alzheimer’s struck Hank, suddenly the Newman-Kates family felt powerless. Together, they valiantly fought the disease until Hank’s death in 2016 at age 77. For years, Dr. Newman has been an ardent supporter of Alzheimer’s research and ADDF, serving on the Board of Overseers since 2013. In recognition of her dedication, we recently honored Pamela with the Charles Evans Award at the Tenth Annual “Hope on the Horizon” Fall Symposium & Luncheon in New York City.
Developing Drugs
December 10, 2019
Stepping Forward in Alzheimer’s Disease Research: A Combination Therapy Approach
Chief Medical Officer at Amylyx Pharmaceuticals gives a closer look at how far they have come in their recently expanded phase 2 clinical trial, supported in part by the ADDF, which is studying a combination approach to treating people with Alzheimer’s disease.


ADDF Impact
November 14, 2019
Hope of the Horizon: Repurposing Drugs for Alzheimer’s
ADDF’s Annual “Hope on the Horizon” Symposium featured a panel of highly esteemed scientists dedicated to Alzheimer’s research.


ADDF Impact
September 24, 2019
Award-Winning Scientists Weigh in on New Directions in Alzheimer’s Drug Research
Research panel featuring recipients of the Goodes Prize weigh in on new directions in Alzheimer’s research.


Understanding Dementia
June 6, 2019
LATE: A New Term for Another Type of Dementia
Researchers have identified a new form of dementia — LATE — that usually affects the oldest old individuals.
ADDF Impact
April 16, 2019
PET Scans Improve Management of Patients with Dementia
A national study has found that PET scans significantly influenced clinical management of patients with dementia.


ADDF Impact
January 14, 2019
Top 5 Advances in 2018
In 2018 we have seen increased investments in Alzheimer's biomarkers and increased interest in prevention.


ADDF Impact
November 13, 2018
Leading Scientists Share Exciting Research Developments at the ADDF Fall Symposium
At the ADDF's Ninth Annual Fall Symposium, Richard S. Isaacson, Michelle M. Mielke, and Mark A. Mintun discussed Alzheimer's biomarkers and prevention.


Developing Drugs
September 24, 2018
Two Decades of Research: The ADDF’s 19th International Conference on Alzheimer’s Drug Discovery
Researchers shared updates on their approaches to developing Alzheimer's treatments at our annual conference.


Supporter Stories
September 21, 2018
The Cure Needs You: Getting Involved in Clinical Trials
Marty Reiswig talks about the importance of participating in Alzheimer's clinical trials.


Developing Drugs
August 2, 2018
Top Five Highlights from AAIC 2018
The ADDF looks back at the 2018 Alzheimer's Association International Conference.


Developing Drugs
June 20, 2018
Gene Therapy, A New Frontier in Alzheimer’s
Dr. Ron Crystal and his team at Weill Cornell developed a gene therapy to replace APOE4 with APOE2, which would significantly lower Alzheimer's risk.


Understanding Dementia
June 14, 2018
Familial Alzheimer’s Disease, Explained
Learn about early-onset familial Alzheimer's disease, which can strike people as early as their 30s or 40s.


Supporter Stories
May 11, 2018
Loss and Found
Adam Rowe recounts a harrowing experience with his mother, who has frontotemporal dementia.


Developing Drugs
May 4, 2018
Alzheimer’s Clinical Trials, Explained
Learn about clinical trials, an important and growing part of our funding at the Alzheimer’s Drug Discovery Foundation.


ADDF Impact
March 14, 2018
ADDF Grantees Headline NIH Alzheimer’s Summit
ADDF-supported research was at the forefront of the Summit and the National Plan to Address Alzheimer's.

Understanding Dementia
February 21, 2018
Types of Dementia (It’s Not Just Alzheimer’s)
While Alzheimer's accounts for more than two-thirds of dementia cases, the ADDF also funds research to prevent and treat the other causes of dementia.


ADDF Impact
January 30, 2018
Meeting the Challenge of CTE
For CTE Awareness Day, we highlight our support of diagnostic tools and treatments for this disease.


ADDF Impact
January 2, 2018
Top Five Alzheimer’s Advances in 2017
Our top five advances in 2017 spotlight the substantial progress we have made toward conquering Alzheimer’s and other neurodegenerative diseases.
Developing Drugs
December 28, 2017
Alzheimer’s Biomarkers, Explained
Learn what a biomarker is, why they matter to the ADDF, which biomarkers we have, and which ones we need.


Developing Drugs
November 28, 2017
Closing in on a Cure: Dr. Roberta Diaz Brinton and Allopregnanolone
With funding from the ADDF since 2004, Dr. Roberta Diaz Brinton has developed what may become the first ever regenerative treatment for Alzheimer's disease.


ADDF Impact
October 30, 2017
CTAD Conference Highlights Our Funded Research
Wednesday marked the beginning of National Alzheimer's Disease Awareness Month and the 10th Clinical Trials on Alzheimer's Disease conference, which featured a record amount of research we supported.


Developing Drugs
October 24, 2017
Combination Therapy: The Right Approach for Alzheimer’s
Learn why combination therapy holds such promise for treating Alzheimer's disease, and why Amylyx's drug is the first such therapy we are funding.


ADDF Impact
September 5, 2017
Driving Innovation in Alzheimer’s Therapies
At our 18th Annual Conference on Alzheimer’s Drug Discovery in Jersey City, the impact of our approach will take center stage.


Supporter Stories
August 30, 2017
Stealing Memories
Jeanette Arahood explains how her patients inspired her to join our TCS New York City Marathon team.
ADDF Impact
August 17, 2017
Inflammation, The Driver of Alzheimer’s Disease?
A growing body of evidence suggests that inflammation in the brain is implicated in the development of Alzheimer's.


Understanding Dementia
July 26, 2017
Contact Sports & CTE: Are the Risks Too High?
A new study reveals a high percentage of former football players died with CTE, including 99% of NFL players tested.


Developing Drugs
June 29, 2017
Three Themes Emerge at BIO2017
Dr. Lauren Friedman details the three topics that dominated discussions at this year's BIO International Convention.
Understanding Dementia
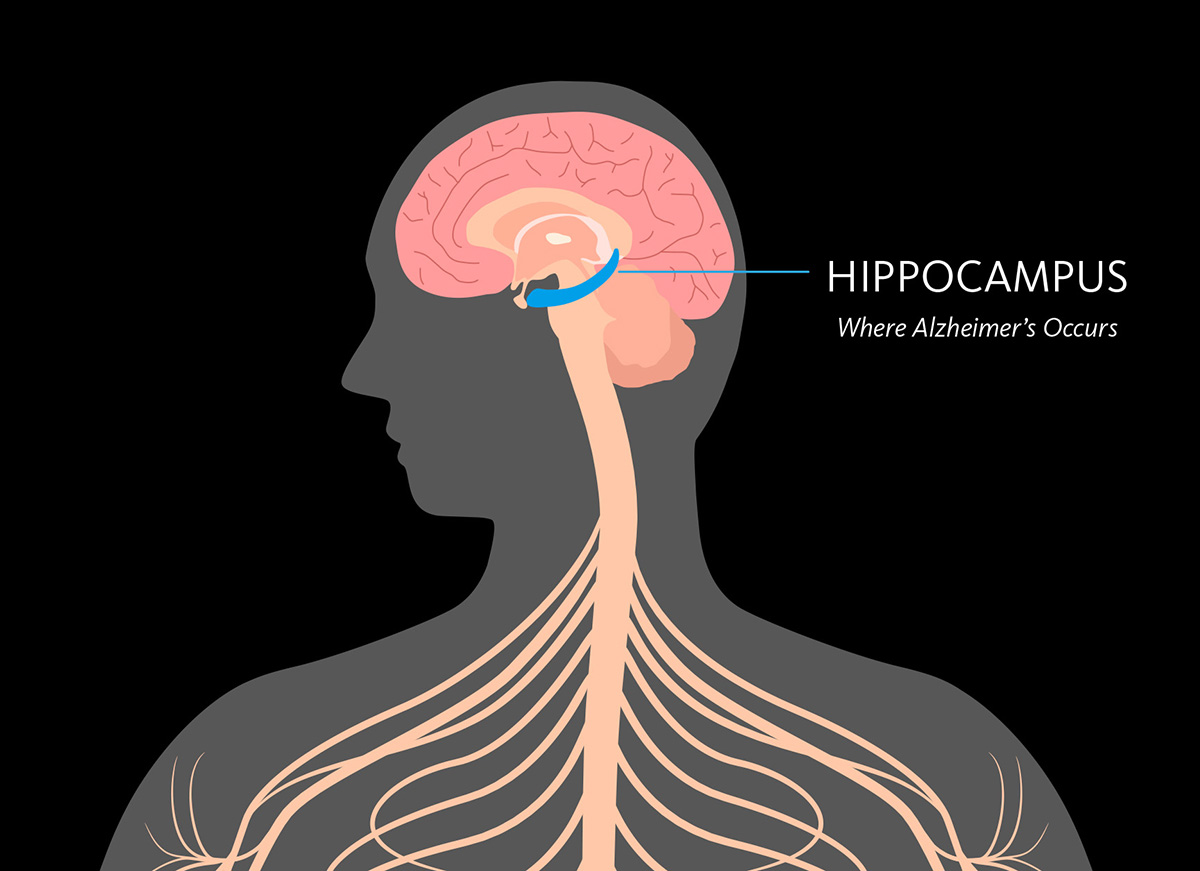
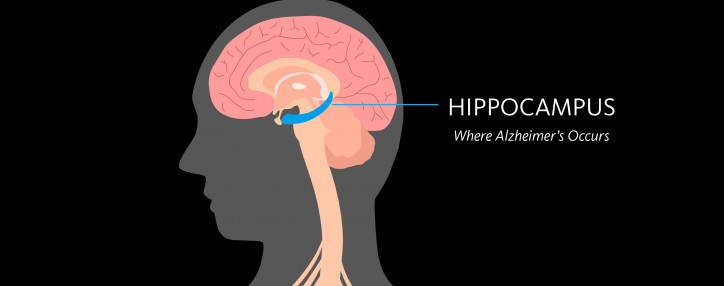
June 20, 2017
Dementia & Alzheimer’s Disease: What’s the Difference?
Our CSO Dr. Fillit explains the differences between Alzheimer's and dementia, two terms often used interchangeably.

ADDF Impact
May 16, 2017
Clinical Trials Inspiring Hope
Since 1998, the ADDF has invested over $100 million in the best ideas to cure Alzheimer's. We are seeing the impact of our efforts, with many promising drugs in clinical trials on the way.


Developing Drugs
April 28, 2017
Trial of First Broad-Spectrum Drug for Neurodegeneration Seeking Volunteers
ADDF grantee Dr. Raymond Scott Turner is recruiting patients for a phase 2 trial of nilotinib—a leukemia drug being tested for treating Alzheimer’s.


Supporter Stories
March 7, 2017
Donor Spotlight: Joyce Cowin
Longtime supporter Joyce Cowin talks about why she has a new sense of hope about Alzheimer's treatments.


Protecting Brain Health
March 6, 2017
Prevention: A Key to Conquering Alzheimer’s
Dr. Howard Fillit explains the importance of prevention to the ADDF's mission and our current efforts, including clinical trials.

Developing Drugs
February 21, 2017
Closing in on a Cure: Dr. Michela Gallagher and AGB101
Dr. Michela Gallagher of Agenebio is developing AGB101, which can potentially prevent mild cognitive impairment from progressing to Alzheimer's disease.
Developing Drugs
February 15, 2017
Does Verubecestat Signal the End of the Amyloid Hypothesis?
Merck announces the end of another anti-amyloid drug trial, calling in to question the amyloid hypothesis and the fate of other anti-amyloid drugs.


Developing Drugs
January 17, 2017
Closing in on a Cure: Oryzon Genomics and ORY-2001
Discover ORY-2001, a revolutionary epigenetic therapy for Alzheimer's disease being developed by Oryzon Genomics in Barcelona, Spain.

ADDF Impact
January 10, 2017
Top Five Alzheimer’s Advances in 2016
In 2016, we made great progress toward finding drugs to prevent and treat Alzheimer’s, as our top five advances demonstrate.


Developing Drugs
December 15, 2016
Closing in on a Cure: Dr. Frank Longo and C-31
We have supported over 20% of all Alzheimer's treatments currently in clinical trials. One of the most promising is C-31, developed by Frank Longo, MD, PhD.


ADDF Impact
November 28, 2016
Striding Toward $100 Million on Giving Tuesday
Since 1998, the ADDF has invested $97 million in critical research and—with your #GivingTuesday donation—we will reach $100 million this year.


Developing Drugs
November 25, 2016
What Does the Failure of Solanezumab Mean?
Eli Lilly announced that its Alzheimer's drug solanezumab failed in its last phase 3 clinical trial. Howard Fillit, MD, discusses what this means, and where we go from here.


ADDF Impact
September 21, 2016
Looking Ahead on World Alzheimer’s Day
On World Alzheimer’s Day, we celebrate the progress made toward finding effective treatments and look toward the future.


Developing Drugs
September 12, 2016
Advancing Epigenetics: A New Approach to Treating Alzheimer’s Disease
Dr. Howard Fillit discusses epigenetic treatments, one of the newest and most promising areas of our portfolio.


Supporter Stories
September 7, 2016
My Marathon Journey with the ADDF
Ernest Scheyder, a returning member of our TCS NYC Marathon team, shares why he is running for Alzheimer’s research and how you can help our dedicated runners.
Developing Drugs
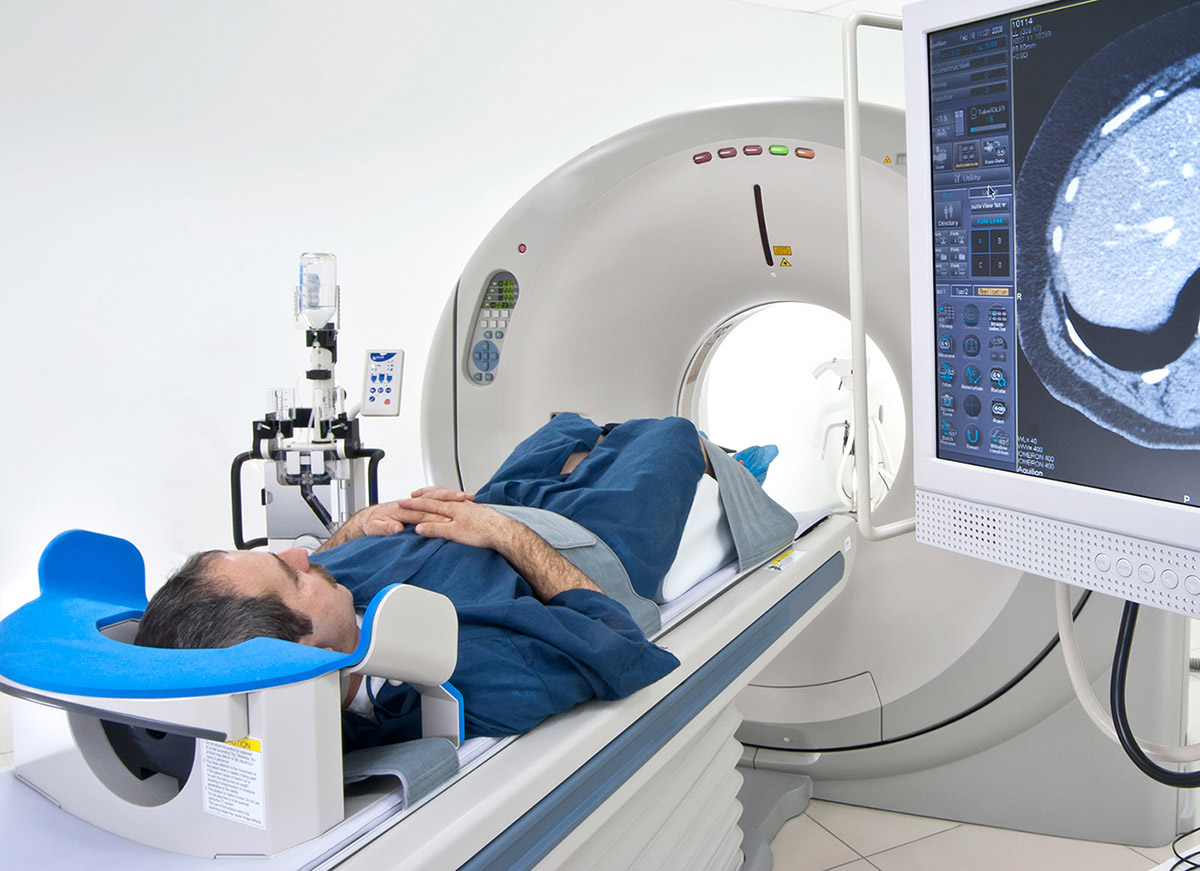
September 2, 2016
ADDF-Funded PET Imaging Makes Clinical Trial Success Possible
A treatment has reduced beta-amyloid plaques in the brains of patients with Alzheimer’s disease.

Supporter Stories
July 6, 2016
The Enduring Legacy of NFL Star Tommy Mason
Karen Mason reflects on her late husband, Tommy Mason, whom she describes as "larger than life," a trait that remained even as he endured the effects of Alzheimer’s.


Developing Drugs
March 1, 2016
Dr. Carmela Abraham Founds New Biotech
With support from the ADDF, Carmela Abraham, PhD recently founded Klogene to accelerate the development of a new class of drugs to combat Alzheimer’s disease.


Developing Drugs
February 25, 2016
Alzheimer’s Drug Featured in Time Was Made Possible by the ADDF
Grantee Dr. Frank Longo's promising new drug to treat Alzheimer's is on the cover of Time magazine.


ADDF Impact
January 11, 2016
2015: Our Most Successful Year
Executive Director Howard Fillit, MD, looks back at 2015—our most successful year ever—and thanks the supporters and scientists who made it possible.

Supporter Stories
December 18, 2015
The Richness of Memory
Adam Rowe shares his experience planning his parents' 50th anniversary while accommodating limitations caused by his mother's frontotemporal dementia.


ADDF Impact
December 1, 2015
Giving Tuesday
For #GivingTuesday 2015, we invite you learn more about our critical work to conquer Alzheimer's disease.


Supporter Stories
October 8, 2015
Allison Johnson: A Runner’s Story
Allison Johnson, a member of our TCS New York City Marathon team, shares why she's running to cure Alzheimer's.


Developing Drugs
June 10, 2015
Investing in a Cure
Investing more in drug discovery and development is the best chance we have for finding a cure for Alzheimer's disease.

Supporter Stories
May 8, 2015
A Mother’s Voice
Adam Rowe reminsces about his mother's voice, the devastating feeling when Alzheimer's disease quiets it. Or silences it completely.

Developing Drugs
April 30, 2015
Partners for a Cure: Association for Frontotemporal Degeneration
Partnerships increase our funding power and our combined expertise—ultimately, ensuring that we can get to a cure for Alzheimer’s and related dementias faster.


Protecting Brain Health
April 6, 2015
Parents Need Answers About Youth Sports Concussions
As a parent, it wouldn't take much evidence for me to keep my children out of high-impact sports like football and soccer. But as a scientist and a physician, I have a different perspective.


Protecting Brain Health
March 20, 2015
Does a Head Injury Increase Your Dementia Risk?
As part of Brain Injury Awareness Month, we’re shining a spotlight on research into the link between Alzheimer’s disease and traumatic brain injury.


Understanding Dementia
March 16, 2015
6 Things to Know About Alzheimer’s
Dr. Roberta Diaz Brinton and Dr. Howard Fillit discuss the latest in Alzheimer’s research and drug development in a recent episode of Alzheimer’s Talks.

Supporter Stories
March 5, 2015
Love is Not Lost
Loss is a natural part of caring for an Alzheimer's patient. These brave men and women facing Alzheimer's have lost many things in their lives. Love isn't one of them.
Developing Drugs
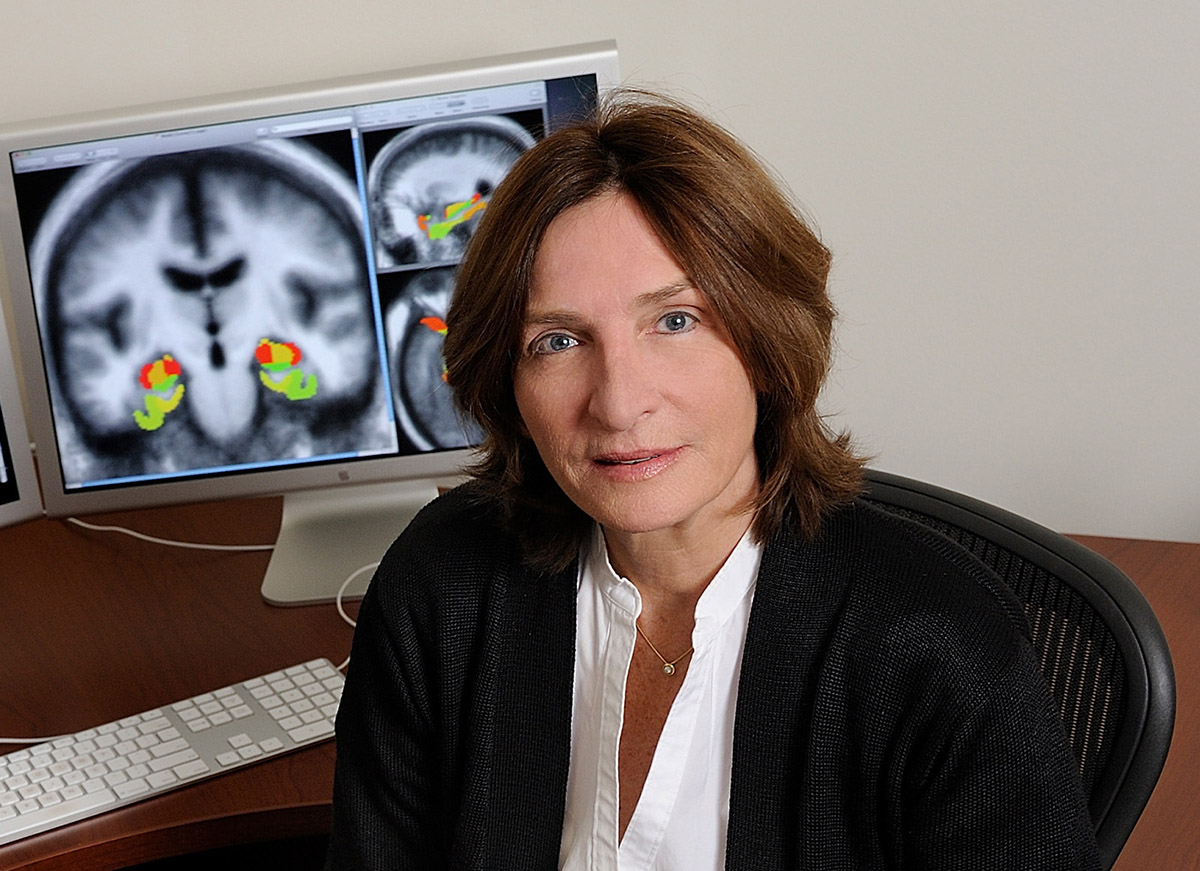
February 3, 2015
Early-Stage Dementia Treatment Headed to Phase 3 Clinical Trial
ADDF-funded researchers are preparing for a phase 3 trial of what could become the first drug approved for mild cognitive impairment.


ADDF Impact
January 15, 2015
A Banner Year for Alzheimer’s Drug Discovery
In 2014, we had a transformative year, advancing drugs for Alzheimer's, supporting the best science, and ensuring people have information to protect their brain health.

Developing Drugs
December 18, 2014
Could an Erectile Dysfunction Drug Treat Dementia?
The ADDF and the Alzheimer’s Society UK have teamed up to support a trial of an erectile dysfunction drug as a possible treatment for vascular dementia.


Understanding Dementia
December 10, 2014
Living with Alzheimer’s: Tips for a Happy and Healthy Holiday Season
It’s no secret that the holidays can be a stressful time. For families living with Alzheimer’s, that stress is often magnified.


ADDF Impact
December 4, 2014
The $3.3 Billion Case for Venture Philanthropy
The Cystic Fibrosis Foundation received a record-breaking $3.3 billion return on its early investment in research that led to the first FDA-approved drug to treat the causes of cystic fibrosis.


ADDF Impact
November 26, 2014
3 Reasons We’re Thankful
This Thanksgiving, we are grateful for the support of our donors, which has enabled the ADDF to make tremendous progress in our search for a cure for Alzheimer’s disease.


Supporter Stories
November 18, 2014
Dan Gasby: “I’m Fighting to End Alzheimer’s”
Four years ago, I received the shock of a lifetime: my wife of 22 years, B. Smith, was diagnosed with early-onset Alzheimer’s disease.


Understanding Dementia
October 20, 2014
A Three-Dimensional Model of Alzheimer’s
Researchers at Massachusetts General Hospital successfully created a laboratory model of Alzheimer's, which may be a more accurate way to screen prospective drugs.
Understanding Dementia
October 9, 2014
What Does the Nobel Discovery Mean for Alzheimer’s Disease?
John O’Keefe, May-Britt Moser, and Edvard Moser, have been awarded the 2014 Nobel Prize in Physiology or Medicine for their discoveries of place and grid cells in the brain.


Supporter Stories
October 7, 2014
Meet Our Marathon Team Member: Dr. Rachel Lane
Rachel Lane, PhD, shares her passion for Alzheimer's disease research and her progress in training for the TCS NYC Marathon.


Supporter Stories
October 6, 2014
Join the Fight to End Alzheimer’s
Leonard A. Lauder explains why he and his brother, Ronald, co-founded the ADDF, and why they need your help to fulfill its mission of conquering Alzheimer's disease.

Protecting Brain Health
October 2, 2014
Lithium & Your Brain Health
Some research shows an association between lithium exposure and "beneficial" outcomes. Are the benefits enough to warrant widespread use?


Developing Drugs
September 24, 2014
New Drug Targets Are Highlight of Alzheimer’s Conference
Presenters at our recent conference shared exciting progress on new Alzheimer's drug targets and biomarkers.


Developing Drugs
August 18, 2014
Drugs to “Fix” the APOE4 Gene May Be Safe
New research reveals that a person without the APOE gene—which influences Alzheimer’s risk—can function normally, paving the way for gene therapies.


Protecting Brain Health
August 11, 2014
Alzheimer’s Disease is the Greatest Threat to Healthy Aging
By 2050, more than 1.5 billion people worldwide will be over the age of 65. Alzheimer's threatens both their health and productivity.


Understanding Dementia
July 28, 2014
5 Highlights from the Largest Alzheimer’s Conference
Our neuroscientists share highlights from their favorite sessions at the 2014 Alzheimer’s Association International Conference (AAIC) in Copenhagen, Denmark.


Understanding Dementia
July 22, 2014
New Drug Offers Promise for Alzheimer’s
Researchers have identified a promising new drug that prevents abnormal blood clots in the brain, reduces cerebral inflammation, and improves memory in preclinical studies.


Understanding Dementia
July 17, 2014
Alzheimer’s Versus Normal Cognitive Aging
The widespread misconception that Alzheimer's disease is a typical part of aging has serious ramifications for people's health and research into a cure.


Supporter Stories
June 30, 2014
An Alzheimer’s Widow Finds Hope in Drug Discovery
My husband and I had been together 26 years when he became one of nearly six million North Americans diagnosed with Alzheimer’s disease. Seven years later, I was an Alzheimer’s widow.


ADDF Impact
June 27, 2014
Joining Forces to End Alzheimer’s
We joined the Accelerating Medicines Partnership to speed up drug development for Alzheimer's and other major diseases.


Protecting Brain Health
June 18, 2014
The Psychological Risk Factors for Dementia
Are cynical people more likely to develop dementia? A new study in Neurology examines the surprising link between cynicism and dementia.


Supporter Stories
June 12, 2014
My Father’s Battle with Alzheimer’s Disease
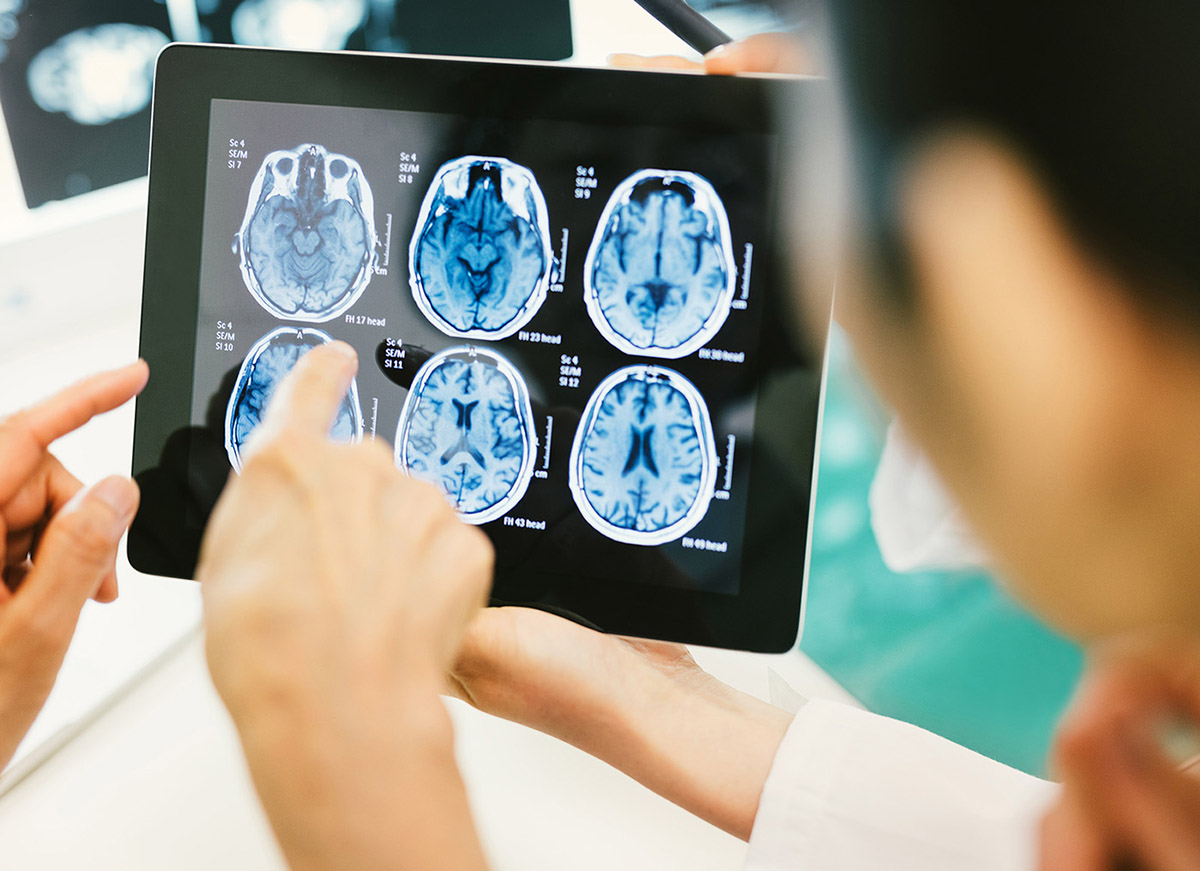
Like many people experiencing Alzheimer's in its early stages, my father began misplacing important objects and forgetting the names of people.
Developing Drugs
June 10, 2014
Virtual Biotechs Maximize Lean Drug Discovery Budgets
Investing in "virtual biotechs" is an efficient way to fund high-risk and high-reward Alzheimer's research.


Developing Drugs
June 5, 2014
An Alzheimer’s Treatment in Your Medicine Cabinet?
A recent New York Times Op-Ed highlights that, while it's possible to "repurpose" available drugs to treat patients, we don't test them because "no one stands to make money."


Developing Drugs
May 23, 2014
Will the First Alzheimer’s Treatment Be a Repurposed Drug?
Dr. Jeffrey Cummings talks about the promise repurposed drugs offer to people with Alzheimer's and other dementias.


Understanding Dementia
May 21, 2014
Q&A on Women and Alzheimer’s Disease
The ADDF's Dr. Howard Fillit answers questions about Alzheimer's during the 4th Annual Great Ladies Luncheon and Fashion Show.


Protecting Brain Health
May 20, 2014
The Case for Dementia Screening
Healthcare professionals rarely screen patients—even those with many risk factors—for cognitive issues or dementia.


Understanding Dementia
May 8, 2014
Understanding the Biology of Aging
Aging is the single greatest risk factor for Alzheimer’s disease. The challenge is translating this knowledge into a cure for the disease.
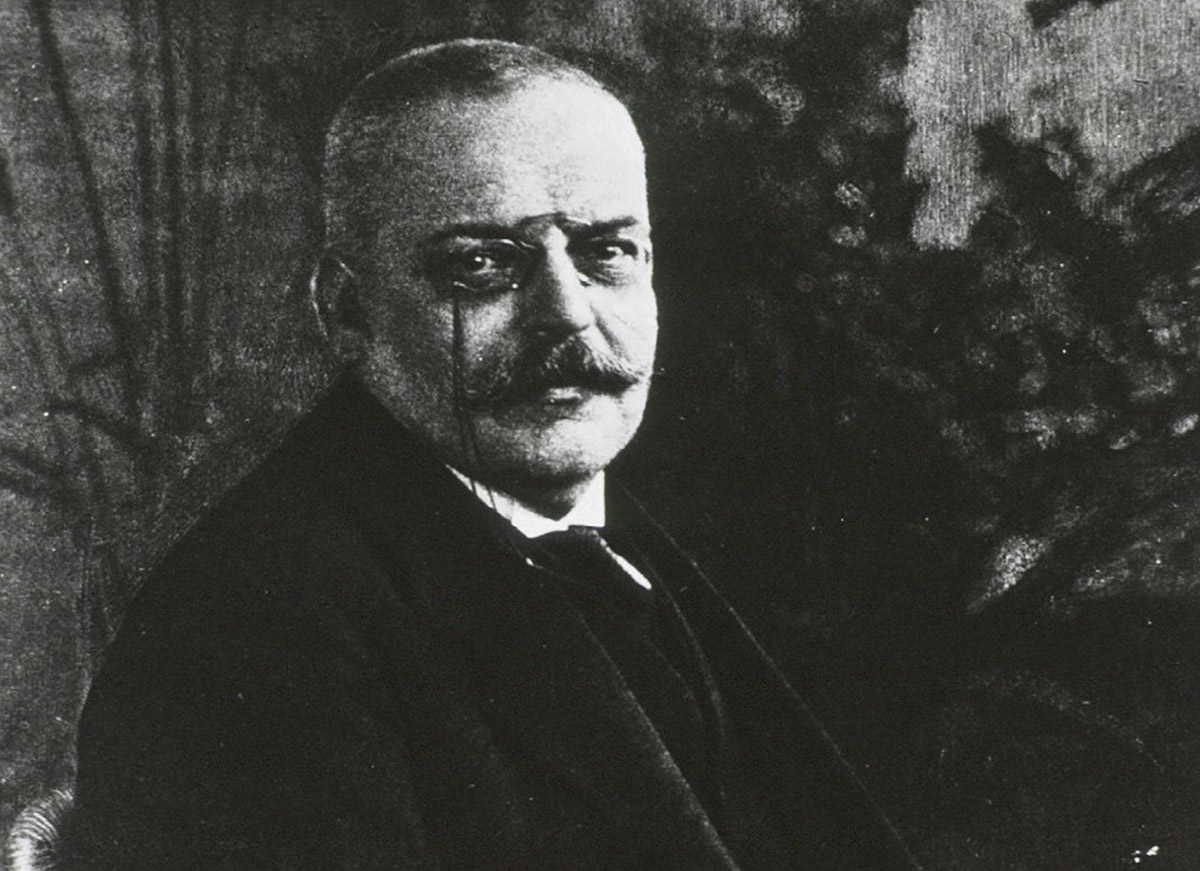
Understanding Dementia
May 6, 2014
Then & Now: Alzheimer’s Research
As a medical student in the 1970s, I studied hundreds of diseases. But one never even got a mention: Alzheimer’s.


Protecting Brain Health
April 22, 2014
Manage Alzheimer’s Risk Factors To Lower Costs
A study from Tufts Medical Center adds to the growing body of evidence suggesting that it is possible to lower one’s risk of developing Alzheimer’s disease.


Understanding Dementia
March 28, 2014
Alzheimer’s Disease: An Unrecognized Killer
We've known for years that the number of deaths caused by Alzheimer's disease has been underreported. A recent study suggests it may be the third-leading cause of death in the U.S.


ADDF Impact
March 1, 2014
2013: A Year of Bold New Initiatives and Clinical Trials
In 2013, the ADDF funded nearly 40 drug research programs around the world. We look forward to continuing to advance the most promising Alzheimer’s research and improving the lives of millions around the world.
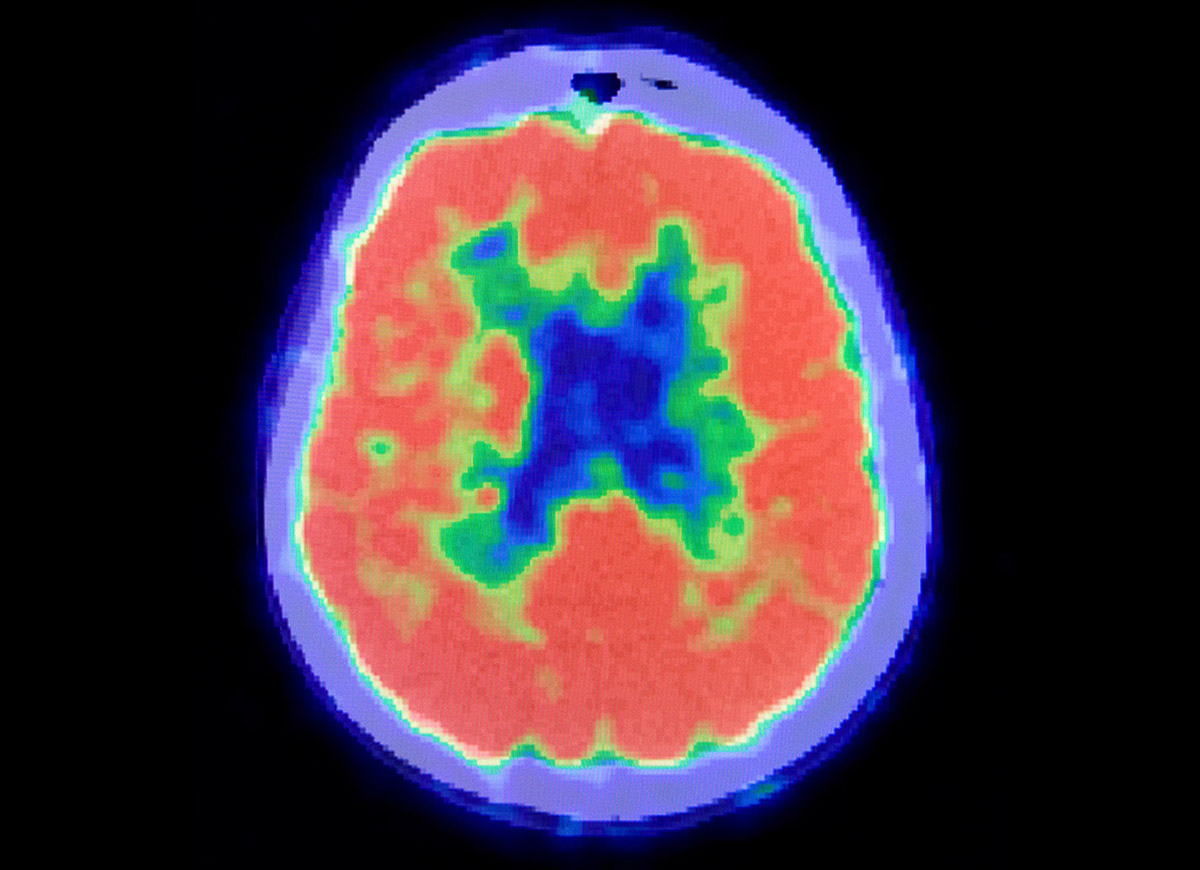
ADDF Impact
August 19, 2013
The Balancing Act of Healthcare Reform
I recently saw a patient who was afraid he had Alzheimer's disease. A new scan made it easy to give him an accurate diagnosis. So why can't most patients use it?
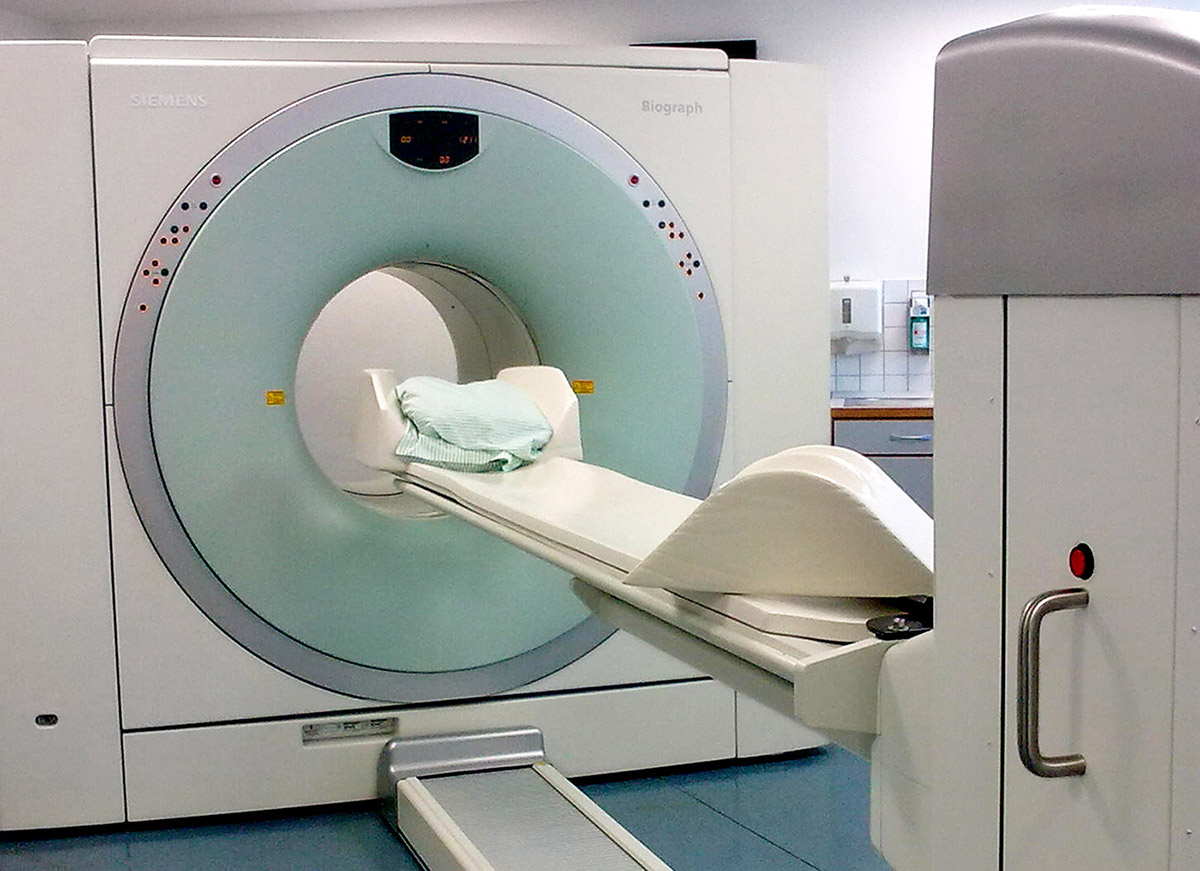
Developing Drugs
June 4, 2013
Fighting Alzheimer’s Costs Now & in the Future
The current costs of dementia care are enormous. But the future costs could create a crippling financial burden on our society, as the number of people diagnosed with dementia is expected to more than double in the next 30 years.
New York, NY 10019
info@alzdiscovery.org
212.901.8000
© Copyright 2026. The Alzheimer's Drug Discovery Foundation, a not-for-profit, section 501 (c)(3).

